|
Introduction: An experiment to show how the Metis R&D Battery Safety Sensor can be used to detect an increase in pressure and explosive levels of hydrogen in a battery enclosure following the first venting phase of a single cell, informing decisions that can prevent the second venting event and thermal runaway of the whole battery system. Setup: To run the experiment and obtain the data, we used the following equipment:
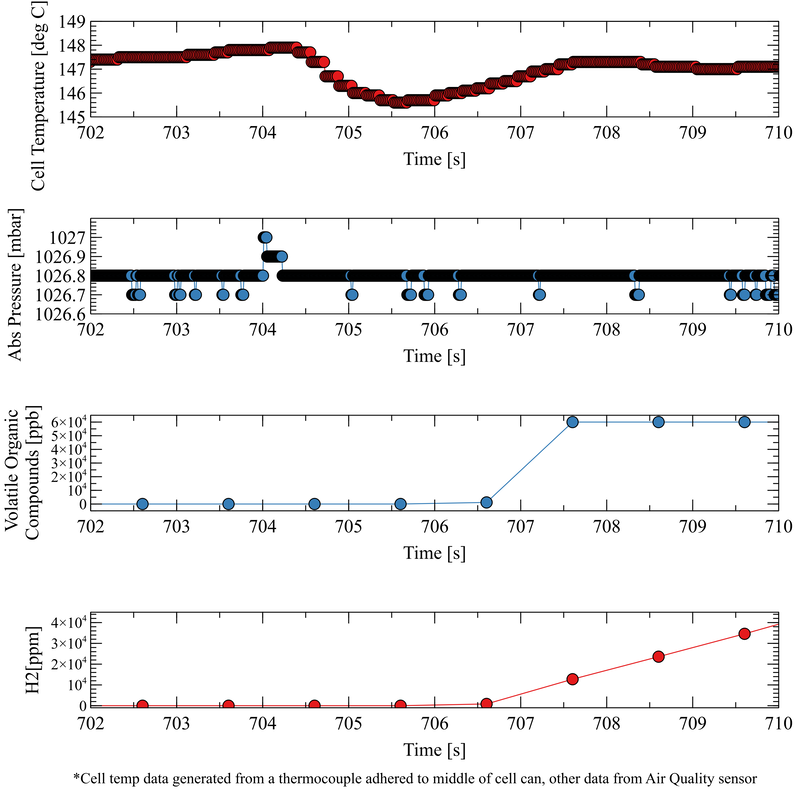
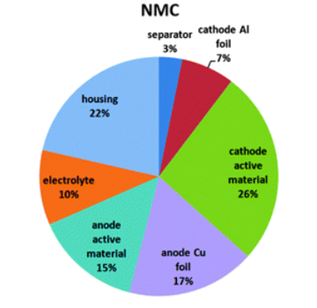
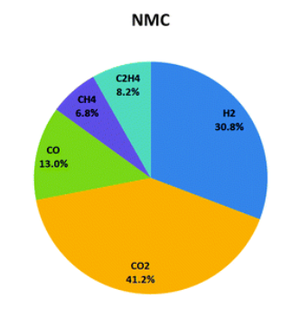
Data Visualisation: To imitate the elevated cell temperature that in real life scenarios might be caused by short circuit, overcharge, over-discharge, external heating or self-heating, a heating pad was wrapped around the cell to directly apply heat. A thermocouple was attached to show the external cell temperature. After approx. 11 minutes a cell temperature of 148 oC was reached and the cell began to visibly vent hazardous electrolyte and associated gases into the Perspex box used to imitate the air tight containment of a battery enclosure. Similarly, in the study: ‘Thermal-runaway experiments on consumer Li-ion batteries with metal-oxide and olvin-type cathodes’, the first vent, or the opening of the safety vent device of the battery housing occurred at approximately 160 oC*. The venting was detected immediately by the Battery Safety Sensor’s precision pressure sensor, the data from which is shown in the middle graph, below. A few seconds later, the venting was also detected through the rapid increase in Volatile Organic Compounds and Hydrogen – see the bottom two graphs. The application of heat to the cell was immediately ceased following the observation of the first venting event, preventing the cell reaching the temperature that would have triggered the second venting event or rapid thermal runaway. Background: The typical mass percentage of each component of a Lithium ion battery with a cathode of Nickel Manganese and Cobalt – Li(NiMnCo)O2 is as follows: The electrolyte consists of dimethyl carbonate (DMC), ethyl methyl carbonate (EMC), ethylene carbonate (EC) and propylene carbonate (PC) in the following mol ratio: DMC : EMC : EC : PC (7 : 1 : 1 : 1)*. The gaseous product of a venting LiNMC cell was analysed by gas chromatography in a similar study and was shown to have the following composition: The Metis battery venting experiment only saw the cell reach the first phase of venting, not the second major phase in which a larger proportion of gas is produced. However, the R&D Battery Safety CAN Sensor measured a concentration of 8.6% hydrogen which is a crucial measurement as it is above hydrogen’s Lower Explosive Limit of 4% and thus presents an explosion risk. With this information, actions can be taken to prevent the cell going into thermal runaway. Next Steps: The Metis R&D Battery Safety CAN Sensor also includes a wide ranging pressure sensor. To demonstrate the function of this, the next experiment will involve the observation of pressure seen by a battery enclosure following battery failure, to determine if the battery enclosure structure has been compromised.
References: * Thermal-runaway experiments on consumer Li-ion batteries with metal-oxide and olivin-type cathodes - RSC Advances (RSC Publishing) DOI:10.1039/C3RA45748F
0 Comments
Leave a Reply. |
|
For more information and pricing, please email
info@metisengineering.com |
© Metis Engineering Ltd, 2023
|




 RSS Feed
RSS Feed
